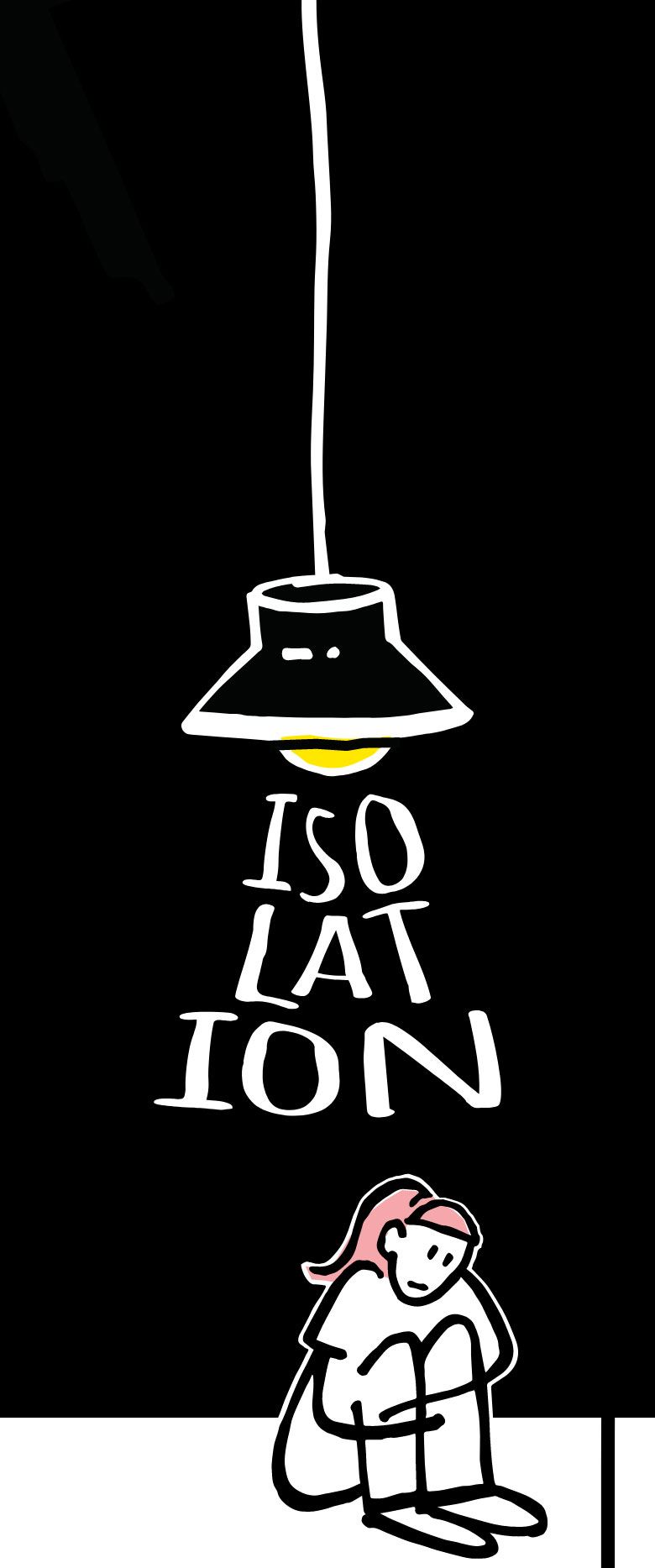
you’re not
actually alone.
Isolation is a common reaction to a traumatic event. Pulling away from people can keep a person with PTSD in an environment that they control. By yourself at home, you don’t have to be on edge about what may happen, and you don’t have to be reminded of how some things are no longer fun for you anymore. This can feel safer. But extended isolation can drive or worsen depression, anxiety disorders, and other mental illness.
Isolation is a common reaction to a traumatic event. Pulling away from people can keep a person with PTSD in an environment that they control.
Take the next steps
When trauma haunts.
Most people don’t complain of regular nightmares, but somewhere between 71% and 96% of trauma survivors with PTSD get them. This often looks like waking up in the middle of the night, overrun with emotions like terror or rage—even sadness. You may feel powerless, and so do people close to you.
Most people don't complain of regular nightmares, but somewhere between 71% and 96% of trauma survirors with PTSD get them.
take the next steps.
Women are diagnosed nearly twice as often as men.
Of people develop PTSD at some point in their lives.
take the next steps.


trauma keeps you stuck.
Hypervigilance is a term that resonates with a lot of people with PTSD. Trauma can leave you constantly waiting for the next thing to go wrong. This is hypervigilance, where you become very sensitive to what’s going on around you and constantly alert. People often become very defensive and otherwise hard to engage with. It can be extremely difficult to stop being hypervigilant. More PTSD treatment options are needed to help stop it – and the other symptoms of PTSD.
Hypervigilance is a term that resonates with a lot of people with PTSD. Trauma can leave you constantly waiting for the next thing to go wrong.
Take the Next Steps.


how study
matching works
To join a study you have to meet its requirements, which are approved by the FDA for everyone’s safety. We’ll use a survey to match you to a study, where a quick screening call will check that you can join. If you’re eligible, you can enroll!
1. Take a quick survey
We’d like to learn more about you in order to find a study that’s a good fit. Studies may have different age or health requirements.
2. Get matched to a study
Based on the information you give us, we’ll try to match you to a suitable study in your area. If we can’t find a good match for you, we’ll keep you in mind for future studies and notify you when they become available.
3. Schedule a screening visit – in person or from your home
The study team will ask questions and check your health to make sure that the study is right for you. This visit can be conducted in person at the study clinic or by an online video call from the comfort of your home.
4. Make study visits
If you join a study, you may be asked to make visits to the study clinic or have online video visits. You may receive an investigational (experimental) vaccine or drug as part of your participation.
Remember, telehealth visits are available.

More About
Clinical Research
Clinical research studies are research projects done with human volunteers to find out if new vaccines and medications are safe and effective. In clinical research studies, the participants get an investigational vaccine or treatment under the care of a doctor and other medical professionals. The word “investigational” means it isn’t available or approved yet for the regular public.
Before any drug or vaccine may be prescribed or administered, it must first go through rigorous testing to show that it is safe and effective for use toward a particular health condition. This testing is referred to as “clinical research.”
Clinical research is closely monitored by government agencies, like the Food and Drug Administration (FDA) in the United States. These agencies must approve any research study before it can even start to enroll participants. Agencies like the FDA have rigorous guidelines that must be adhered to for these studies, including what sorts of studies are allowed, how the studies must be performed and what must be documented, as well as what precautions must be taken to ensure the safety and privacy of all study participants.
Prior to starting a study, its plan—known as a protocol—is reviewed by a panel of science and ethics experts who examine the scientific rationale behind the study while also reviewing how it protects the welfare of its participants. Part of this scrutiny includes making sure the confidentiality of all participants and their personal information is protected.
Even after a study protocol has been approved, these agencies keep a close watch to make sure the protocol’s rules and procedures are adhered to. Once the study ends, the data gathered is analyzed and reviewed before being presented to the government agency. From there, the government agency may decide whether they can safely approve a drug, or if they need more information. The goal of the government agency’s review is to protect the public, and so they will not approve a drug, treatment, or vaccine unless they are confident that it works safely and effective.
Clinical research studies cannot occur without people joining them. But the reasons why people choose to participate in clinical research vary with each individual. Only you can decide that participation is the right choice for you. Talk to your doctor if you’re interested in participating, and bring any questions to your screening visit.

COVID-19
We Remain Open
For the safety of both staff and patients, we require that our research sites are COVID-19 SMART facilities. That means they use best practices to reduce virus exposure and are dedicated to ensuring they remain open and available for the research of investigational medications.
COVID-19 SMART steps we are taking to ensure safety include:
- Greatly increased availability of video office visits
- Ensuring social distancing measures are taken in all waiting areas
- Continuing the practice of cleaning and disinfecting exams rooms thoroughly between every patient visit
- Appropriate personal protective equipment (PPE) for all staff members at our health clinics



Our Trusted PArtners

AcurianHealth helps connect people with clinical research studies that offer treatments under development. We carefully match thousands of people to hundreds of research studies all over the world. As part of AES, a global company that enrolls and engages patients in clinical trials. AcurainHealth is dedicated to helping advance health research.

Synexus is dedicated to conducting clinical studies and has been investigating the effectiveness of new medicines and treatments for more than 20 years. We provide a friendly, relaxed environment where you have the chance to help shape the future of health for yourself and for others. Synexus has study clinics around the world, each with its own dedicated team of highly trained doctors and nurses. Our friendly staff and personal touches make our clinics welcoming places for you to take part in a clinical study.

PPD is a leading global contract organization providing comprehensive, integrated drug development, laboratory and lifecycle management services. Our customers include pharmaceutical, biotechnology, medical device, academic and government organizations. With offices in 47 countries and more than 26,000 professionals worldwide, PPD’s mission is to help our customers deliver life-changing therapies to improve health.